Capitan Orthopedics
Filling the Gap in Rotator Cuff Tear Treatment
The Problem
The Clinical Gap
For decades, patients with irreparable rotator cuff tears have faced a frustrating reality: few options exist to restore movement and relieve pain. Traditional soft tissue procedures are not possible when the tendon is too damaged or retracted, and for most patients under 65, a reverse shoulder replacement isn’t an option. This is what led us to create SupraSpacer™.
Introducing SupraSpacer™
SupraSpacer™ Simple, Reproducible Procedure
SupraSpacer™ acts as a supportive spacer within the shoulder joint. When the rotator cuff is torn and unable to support the joint effectively, the humerus slowly shifts upward, which limits function. By re-establishing normal spacing, SupraSpacer™ realigns the joint to its natural position.
- 7 Surgical Steps
- Minimally Invasive, Bone-Sparing Procedure
- Single-Use, Sterile Packaged Instruments, Trials and Implant

Not Yet FDA Cleared
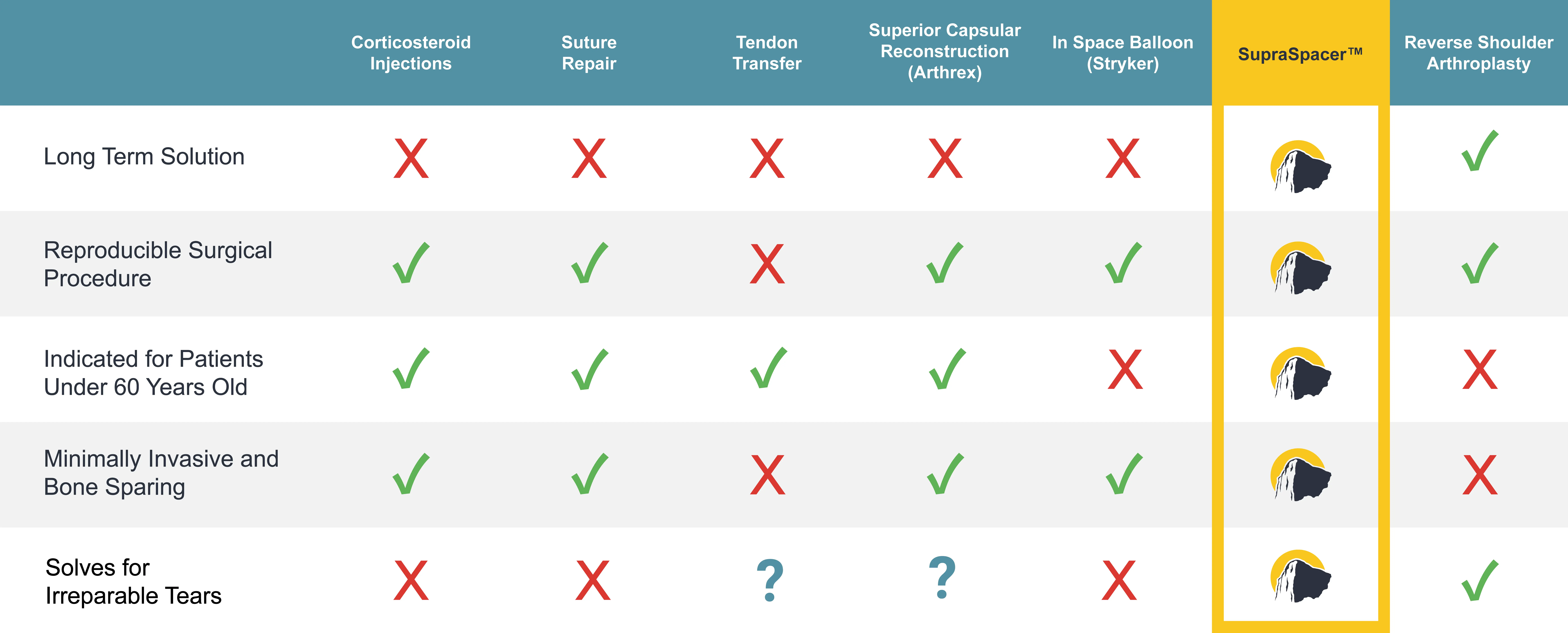
Normal Spacing
A normal glenohumeral joint maintains proper alignment and consistent joint spacing. The humeral head remains centered within the socket, preserving smooth motion and stable shoulder function.
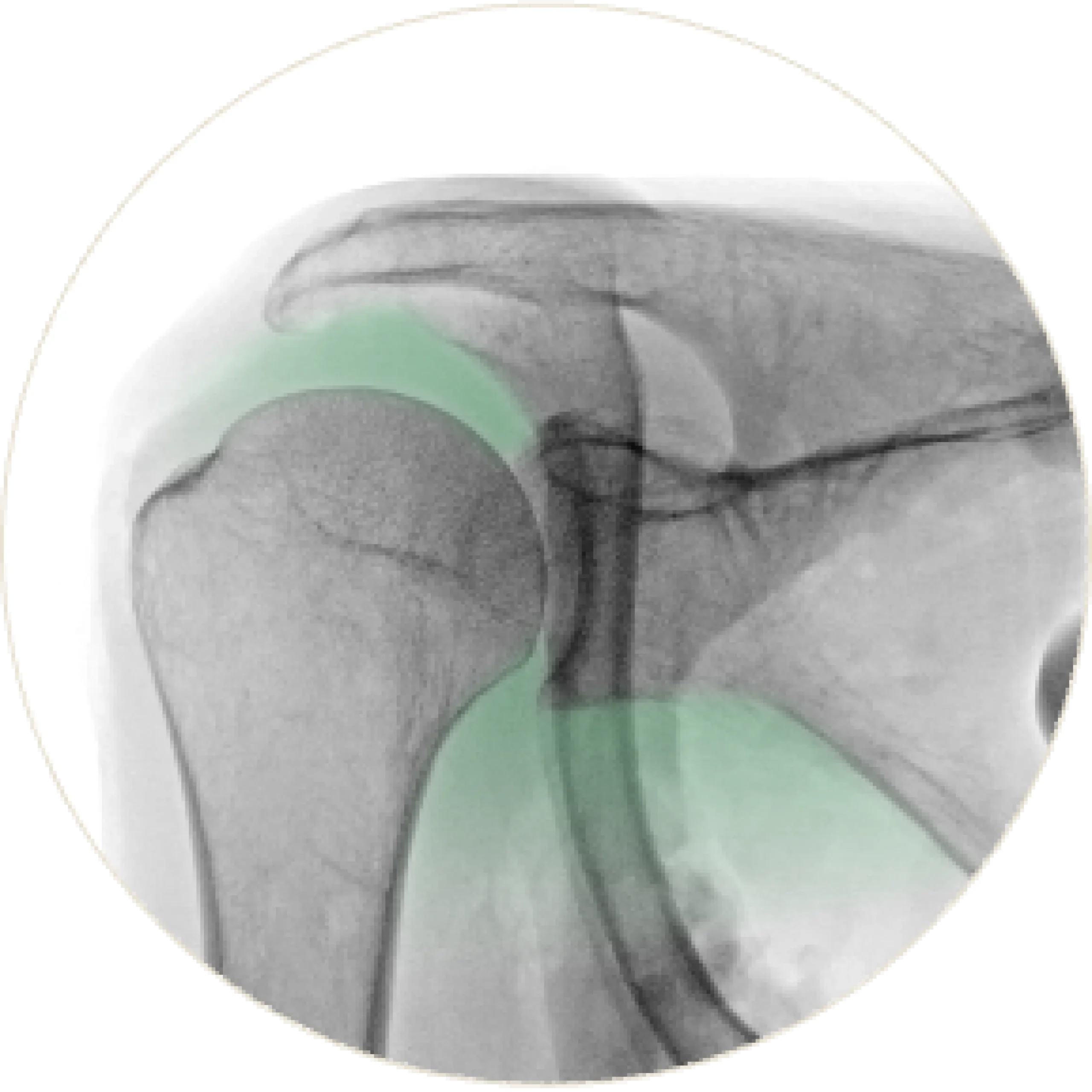
Irreparable Cuff Tear
A irreparable tear causes the humerus to migrate proximally, disrupting natural alignment. This leads to impingement pain, reduced range of motion, and diminished shoulder stability.
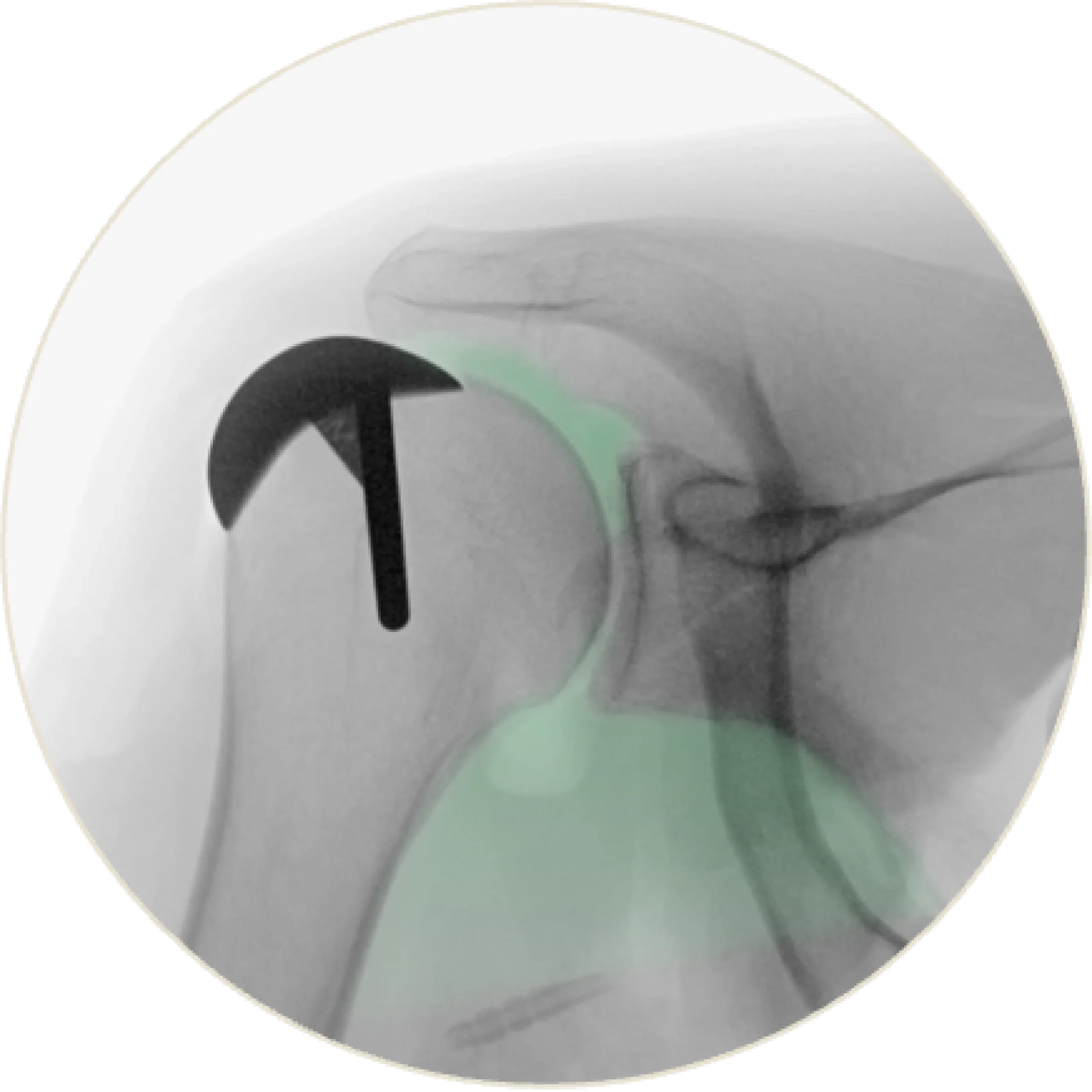
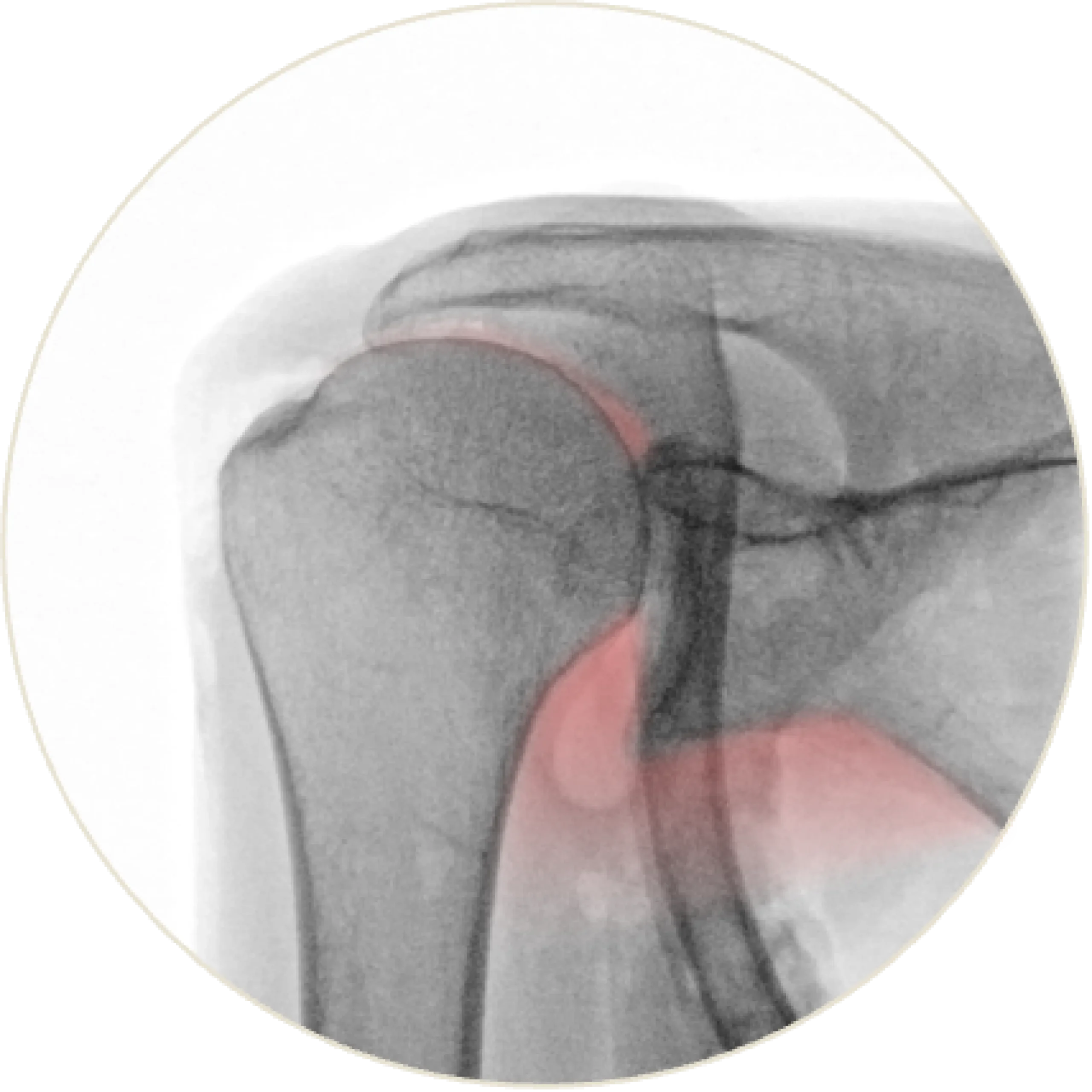
SupraSpacer™
The SupraSpacer™ restores anatomical spacing and natural joint alignment, allowing smooth articulation. It reduces impingement-related discomfort and supports balanced shoulder mechanics.
Who We Are
Surgeon Design Team

R. Sean Churchill, MD, MBA
Shoulder Reconstruction Specialist
Bio
R. Sean Churchill, MD, MBA, serves as Managing Director of the cultivate(MD) Venture Capital Fund and is a Board Member of Genesis Innovation Group, Inc. and Capitan Orthopedics, Inc.. He brings a rare combination of clinical excellence, engineering expertise, and business acumen to the advancement of medical innovation. With more than 20 years of experience as a shoulder reconstruction specialist, Dr. Churchill has been at the forefront of orthopedic advancement since completing his formal training.
Dr. Churchill’s early career in chemical engineering provides him a unique approach to clinical problem-solving, enabling him to develop creative, practical, and scalable solutions to complex healthcare challenges. He is the inventor of the first FDA-approved stemless shoulder replacement and currently holds more than 20 issued and pending patents. His extensive innovation experience includes collaboration on 22 implant design teams, with product designs collectively responsible for more than $2 billion in global product sales since 2007.
In addition to his clinical and technical achievements, Dr. Churchill’s MBA provides him with a deep understanding of the business and financial dynamics of healthcare. This dual perspective is central to his leadership at cultivate(MD), where he plays a critical role in identifying, evaluating, and advancing high-potential medical technologies and startups. Dr. Churchill’s vision for cultivate(MD) extends beyond investment alone. He is committed to building a bridge between groundbreaking innovation and real-world clinical application—accelerating commercialization, reducing healthcare costs, and ultimately improving patient outcomes on a global scale.

Chris Jones, MD
Shoulder Reconstruction Specialist
Bio
- Shoulder Reconstruction Specialist
- 5+ Patents Issued and Pending
- Previous Design Team Experience
- 25+ Years in Practice
- 2,000+ Total Shoulder Arthroplasty Procedures

Jessica Churchill, MD
Shoulder Reconstruction Specialist
Bio
- Shoulder Reconstruction Specialist
- Active Academic Surgeon with 24 Publications and 9 National Presentations
- Medical Director of Denver’s only Orthopedic Center of Excellence
- Previous Design Team Experience
Leadership & Project Team

Chance Leonard
Chief Executive Officer
Bio
Chance W. Leonard is the Chief Executive Officer at Genesis Innovation Group, Inc. and its subsidiary company Capitan Orthopedics, Inc. He brings more than 31 years of distinguished leadership experience in the medical device industry and a solidified reputation as a results-driven executive with expertise in orthopedic innovation, commercialization, and strategic growth. He is widely recognized for his deep industry knowledge, extensive relationships, and proven ability to build high-performing organizations that create significant enterprise value. Known for his influential, determined, and passionate leadership style, Mr. Leonard has consistently driven transformational growth across multiple high-profile orthopedic companies.
Most recently, Mr. Leonard served as Chief Commercial Officer at Skeletal Dynamics, where he played a pivotal role in building the company into a trusted global brand and establishing it as the premier upper extremity company worldwide. Under his leadership, Skeletal Dynamics achieved an extraordinary 330% growth in the U.S. market through execution excellence, a robust commercial strategy, integrated marketing and education initiatives, and a focused, organic portfolio development approach—resulting in substantial enterprise value creation.
Previously, Mr. Leonard served as Vice President at Stanmore Implants, where he successfully led the commercialization of the company’s U.S. platform in the highly competitive oncology and limb preservation market. His leadership and the efforts of his team were instrumental in Stanmore Implants’ acquisition by Stryker in April 2016. Earlier in his career, Mr. Leonard held key leadership roles at Zimmer Trauma and Synthes Trauma.

Tyler Braketa
Vice President of Quality
Bio
- 15+ Years Med Device Quality Engineering
- Quality Leadership Roles with Paragon 28, Zynex Medical and Zimmer Biomet Spine

Luke Aram
Senior Engineer
Bio
- 22+ Years with J&J
- 71 Patents
- 10 Product Launches
- Deep Expertise in Articular Joint Implants

Isaac Running
Project Engineer
Bio
- 10+ Years with GIG
- 18 Patents
- 5 Product Launches
- Expertise in Early-Stage Development

Jon Chau
Patent Attorney, Partner at Honigman LLP
Bio
- Experienced Patent Attorney who Advises Clients on Intellectual Property Matters and Patent Strategies

Rich Mazzola
Co-Founder & CFO of Project MedTech
Bio
- Co-Founder and CFO of Project MedTech
- Finance, Operations and Start-Up Consulting to Early-Stage Companies
- Experience in Start-Ups and Private Equity backed Companies
- Maximize Company Value and Growth Opportunities
- Strengthen Relationships with Investors, Strategic Partners, and Non-Dilutive Sources
Our Team Has Proven Results
301
Milestones Achieved
227
Patents Earned
154
510k Clearances
13
Successful Exits
Advancing Care. Empowering Surgeons. Improving Lives.
For Patients
SupraSpacer™ is designed to support natural shoulder mechanics through a minimally invasive outpatient procedure. The approach and implant both emphasize joint preservation, efficient recovery, and a durable, long-term solution.
For Surgeons
SupraSpacer™ provides a simple, consistent technique that integrates smoothly into existing surgical workflows. The system includes sterile-packaged instruments and implants to support procedural efficiency.
For Surgical Settings
SupraSpacer™ fits seamlessly into ASC environments with a straightforward and reproducible technique. Its sterile-packaged components and efficient setup help maintain consistency across procedures.
For Payers
SupraSpacer™ follows a streamlined procedural framework designed to simplify surgical planning and resource use. The approach supports efficiency and alignment with established outpatient care pathways.
Advancing Care. Empowering Surgeons. Improving Lives.
For Patients
SupraSpacer™ is designed to support natural shoulder mechanics through a minimally invasive outpatient procedure. The approach and implant both emphasize joint preservation, efficient recovery, and a durable, long-term solution.
For Surgeons
SupraSpacer™ provides a simple, consistent technique that integrates smoothly into existing surgical workflows. The system includes sterile-packaged instruments and implants to support procedural efficiency.
For Surgical Settings
SupraSpacer™ fits seamlessly into ASC environments with a straightforward and reproducible technique. Its sterile-packaged components and efficient setup help maintain consistency across procedures.
For Payers
SupraSpacer™ follows a streamlined procedural framework designed to simplify surgical planning and resource use. The approach supports efficiency and alignment with established outpatient care pathways.
Get in Touch with Capitan Orthopedics
Contact us with questions or inquiries about Capitan Orthopedics and our latest developments.